Dr. Christopher Taglia did his bariatric surgery fellowship at North Shore University Hospital in Long Island, where the 900-bed hospital had 800 COVID-19 patients not too long ago. He’s a specialist in gastric-bypass issues at Lompoc Valley Medical Center now, but when COVID struck Santa Barbara, he fell into a New York state of mind.
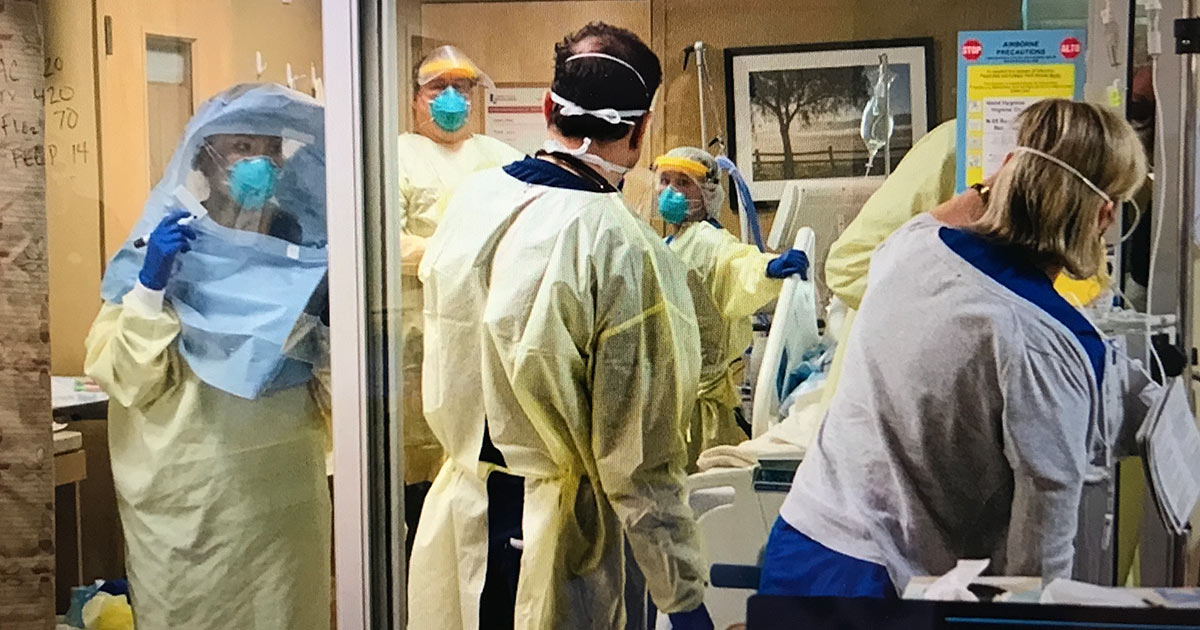
“It was real,” he said. “You’re seeing all these stories of people truly being awesome, going into leadership roles. I wanted to be part of that.” Taglia joined the Lompoc hospital’s Critical Care Unit — he got an excellent education in critical care at Cottage Hospital as a resident, he said — which added the drug remdesivir to its arsenal on May 15.
Santa Barbara County’s three hospital systems are now using remdesivir after more than 100 doses were distributed by the state Department of Health on May 18. The drug works by interfering with the virus’s ability to replicate, but the virus alone isn’t what kills. It provokes an overactive response by the body’s immune system that leads to critical organ failures and death. Giving it to a patient early or later in the course of the disease is the topic of argument now.
Because remdesivir is so scarce, hospitals must take all of that into consideration when determining who gets the medication. But it’s an experimental drug, receiving an FDA emergency-use authorization on the strength of an as-yet-unvetted 1,063-patient study sponsored by the National Institute of Allergy and Infectious Diseases (NIAID). The study showed that patients had a median rate of recovery of 11 days, compared to 15 days for the group of patients on a placebo in a double-blind trial, which means doctors and patients didn’t know who got which.
Wonder Drug

What now seems to be a wonder drug started as a problem child for its manufacturer, Gilead Sciences in the Bay Area. The biopharmaceutical company developed remdesivir in 2009 for hepatitis C and then tried it on Ebola and Marburg viral infections; it proved ineffective for each. Gilead had 1.5 million doses warehoused when clinical trials began for COVID-19 and released them once given the green light by the Food and Drug Administration.
The company’s CEO, Daniel O’Day, announced on April 29 that the drug would be provided gratis for distribution after FDA approval, which came on May 1. The improved recovery rate was good news, but the study’s results on mortality weren’t large enough to be significant, one summary indicated, speculating that treating the disease in the early stages could be of most benefit. While Gilead’s stock prices soared initially on the news, detractors claiming it performed about as well as Tamiflu caused them to sink in recent days.
Because the FDA’s emergency approval for remdesivir lacked the years-long process that drugs normally go through, the peer review is being fought out in medical journals and newsprint. Some critics argue the Gilead-funded trials do not show the cessation of viral replication the company claims. One pointed instead to a trio of drugs — lopinavir–ritonavir, ribavirin, and interferon beta-1b — that stopped the virus from duplicating, as described in a 127-patient study published in Lancet on May 8. Another study in Lancet on May 16 of about 150 patients in China found no benefit from remdesivir.
Precious Meds
The first batch that arrived in Santa Barbara County contained 10 vials, and Lompoc hospital was given a full dose of six vials for a newly arrived patient in critical condition. The rest went to Marian Regional Medical Center in Santa Maria. Dr. J.T. Ritter, an infectious disease specialist at Marian, explained the FDA-recommended treatment takes two vials of remdesivir on the first day and one for four days after that. He said Marian started their first patient on the treatment only three days ago.
Cottage Health created a committee to help make the decision on remdesivir, Dr. Lynn Fitzgibbons said, “and any future COVID-related medication which may be in scarce supply.” She’s the chair of infectious diseases for the three Cottage hospitals and has become known for her calm and factual talks at UC Santa Barbara Economic Forecast COVID webinars.

“The state encouraged hospitals to develop an expert, multi-disciplinary committee to create ethical and evidence-based guidelines for when and how to use remdesivir within hospital systems,” Fitzgibbons explained. “We want to be sure patients who will benefit the most are those who have top priority in accessing this medication.”
“I observed some measurable improvements after the five-day course,” Taglia said carefully, “but unfortunately not enough for extubation.”
Both Fitzgibbons and Taglia said remdesivir’s side effects — described as liver toxicity — had not manifested in their patients. As for whether the drug was working, Fitzgibbons was both positive and cautious: “With such a small number, it’s too early to tell whether there has been definite benefit, but we’re encouraged our patients seem to be doing as well as the clinical trials demonstrate.”
Taglia mentioned a New England Journal of Medicine article that suggested there was a greater benefit for patients who were on supplemental oxygen; in other words, their lungs weren’t yet so compromised that intubation was required. He noted the participants in the NIAID study — which took place at 60 sites in the U.S., Europe, and Asia — received remdesivir on admission and for 10 days. If more of the drug became available, Taglia said, he’d want to consider a 10-day course for patients before they reached the point that oxygen had to be supplied directly down into their lungs.
No study has yet told doctors which patients would end up needing a ventilator, said Taglia, who grew up in N.Y.C. and got his MD at Albert Einstein College of Medicine. “We know that some of the risk factors include obesity, diabetes, and hypertension, but we are not close to predicting this progression with any certainty,” he said. “Obesity seems to be the most significant risk factor we have noticed, which is in line with some of what is being published by the hospitals in New York.”
The State of New York, with a population of 19.4 million, has experienced more than 29,000 deaths to COVID-19. In California, which has twice the population, 3,973 people have died, 12 of them in Santa Barbara County.
At the Santa Barbara Independent, our staff is working around the clock to cover every aspect of this crisis — sorting truth from rumor. Our reporters and editors are asking the tough questions of our public health officials and spreading the word about how we can all help one another. The community needs us — now more than ever — and we need you in order to keep doing the important work we do. Support the Independent by making a direct contribution or with a subscription to Indy+.



You must be logged in to post a comment.